The approved product is bioequivalent and therapeutically equivalent to the reference listed drug, Ravicti Oral Liquid, of Horizon Therapeutics U.S. Holding LLC.
The company said the product will be manufactured at its Unit-III facility and will be launched immediately.
Also read: World leaders have a message for the markets and its not a pleasant one
The drug is indicated for the chronic management of patients with urea cycle disorders (UCDs) who cannot be managed through dietary protein restriction or amino acid supplementation alone.
According to IQVIA data cited by the company, the product has an estimated market size of $50.2 million for the 12 months ending February 2026.
With this approval, Aurobindo Pharma now has a cumulative total of 579 ANDA approvals from the USFDA, including 556 final approvals and 23 tentative approvals.
Earlier this week, Aurobindo Pharma’s subsidiary TheraNym Biologics Pvt. Ltd. expanded its existing contract manufacturing operations agreement with Merck Sharp & Dohme Singapore Trading Pte Ltd to construct and commission a greenfield drug substance manufacturing facility, referred to as Unit 2.
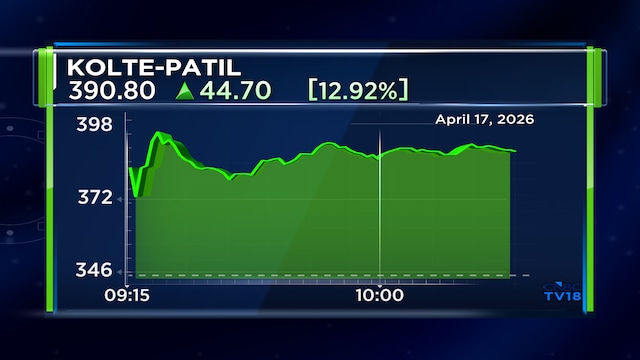
Shares of the pharma major were trading marginally above Thursday’s closing at ₹1,387.30 as of 11.06 am. The stock has gained nearly 8% over the last month and almost 26% in the last six months.
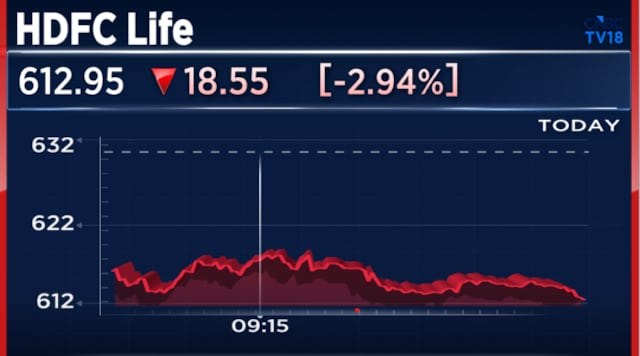
Also read: Here’s why HDFC Life shares are the top Nifty losers after Q4 results