Biocon said the US Food and Drug Administration issued a Form 483, after completing its pre-licence inspection at Biocon’s biosimilars site in Biocon Park, Bengaluru.
The inspection was conducted between April 20 and April 29, 2026 and covered three Biologics manufacturing units, five quality testing laboratories and two warehouses.
Biocon said the observations are procedural and do not pertain to data integrity or quality oversight. No repeat observations were noted, the exchange filing stated.
Biocon said it will submit a comprehensive corrective and preventive action (CAPA) plan within the stipulated timeline and is confident all observations will be addressed fully and expeditiously.
Last week, Biocon said it received approval from Health Canada for two denosumab biosimilars, Bosaya and Vevzuo, which are for the treatment of osteoporosis and cancer-related bone conditions.
The approval was granted on April 3 and it covers Bosaya as a biosimilar to Prolia and Vevzuo as a biosimilar to Xgeva, both in their commonly used dosage forms.
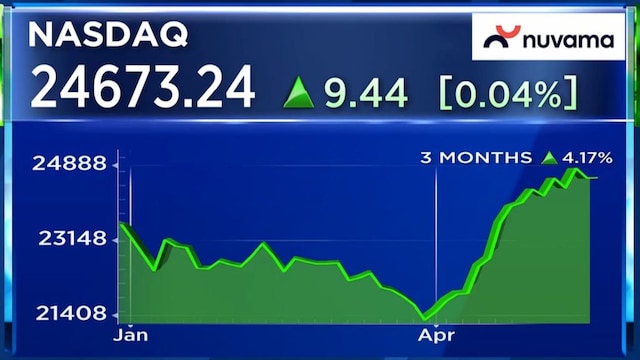
Shares of Biocon are trading 1% lower on Thursday at ₹358.85. The stock is down 8% so far in 2026.